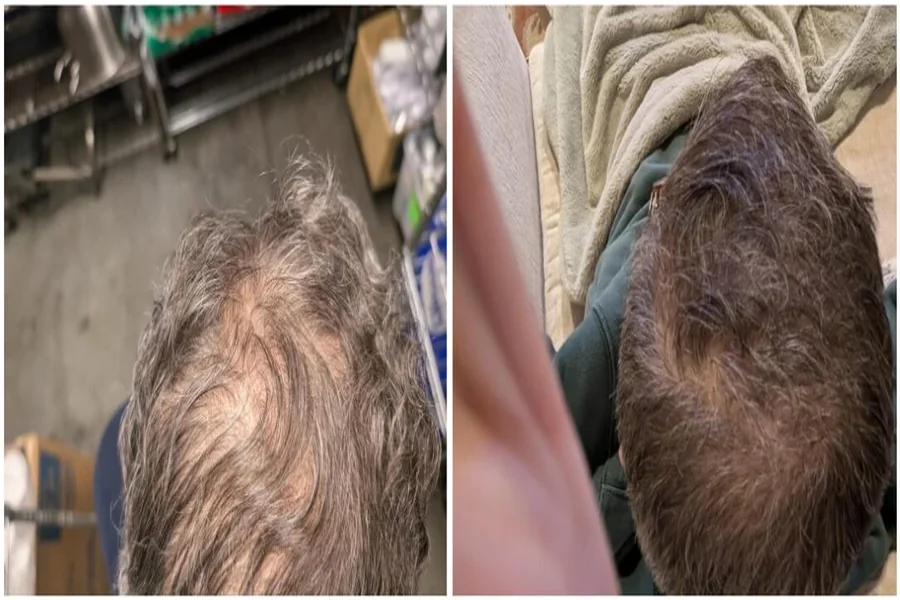
Happy Head’s topical dutasteride has emerged as a promising option for people seeking stronger, thicker hair while minimizing the systemic effects of oral 5-alpha reductase inhibitors. Increasingly available through compounding pharmacies and telehealth clinics, it delivers a potent anti-androgen directly to the scalp where it’s needed most. This article summarizes how topical dutasteride works, reviews the available clinical evidence, outlines common formulations and application protocols, and covers safety, combination strategies, and practical access considerations. It’s written for informed readers who want a concise, evidence-focused perspective on where this therapy fits within a modern hair-loss toolkit.
How Topical Dutasteride Works
Mechanism Of Action: 5-Alpha Reductase Inhibition
Dutasteride is a dual 5-alpha reductase inhibitor: it blocks both type I and type II isoenzymes that convert testosterone to dihydrotestosterone (DHT). DHT is the androgen primarily implicated in androgenetic (pattern) hair loss because it shortens the anagen (growth) phase and miniaturizes susceptible hair follicles. Compared with finasteride, which selectively inhibits type II, dutasteride is pharmacologically more potent and achieves greater suppression of serum DHT when taken orally. The topical strategy is to concentrate dutasteride at the follicular level to reduce local DHT and halt or reverse miniaturization.
Topical Delivery Versus Oral Therapy: Theoretical Advantages
Topical application targets the drug to the scalp while aiming to limit systemic exposure. The hoped-for advantages are similar efficacy to oral therapy but with fewer systemic hormonal effects (for example, lower rates of sexual side effects). Also, topical delivery allows flexible formulations (solutions, gels, foams) and combination with other topical actives. But, achieving consistent scalp penetration and predictable local concentrations remains a formulation and pharmacokinetic challenge.
Who Might Benefit: Patient Profiles And Pattern Hair Loss Types
Topical dutasteride is most relevant for adults with androgenetic alopecia, men with vertex or frontal thinning, and women with diffuse pattern thinning, who either cannot tolerate oral inhibitors or prefer to avoid systemic therapy. It may also appeal to patients who have a partial response to topical minoxidil but need additional anti-androgenic effect. That said, those with non-androgenetic causes (telogen effluvium, scarring alopecia) are unlikely to benefit from a 5-alpha reductase inhibitor alone.
Clinical Evidence And Effectiveness
Key Clinical Trials And Study Findings
Large-scale randomized trials of topical dutasteride remain limited. Several small randomized and open-label studies have evaluated concentrations ranging from 0.01% to 0.5% applied once or twice daily. Many of these studies report statistically significant increases in hair count and improvements in hair thickness versus placebo or baseline, with results comparable to, or in some trials marginally better than, topical finasteride. Notably, a 24-week randomized trial comparing topical dutasteride to topical finasteride and placebo found superior increases in hair count in the dutasteride arm, but sample sizes were small, and follow-up was short.
Expected Timeline For Hair Count And Hair Thickness Improvements
Clinical data and practitioner experience suggest patients may see early improvements (reduced shedding and subjective thickening) within 8–12 weeks, with more measurable increases in hair count and shaft diameter at 4–6 months. Maximum responses typically continue to develop through 12 months. As with other hair therapies, individual variability is substantial and consistent; long-term use is usually necessary to maintain gains.
Limitations Of Current Evidence And Research Gaps
Existing studies are limited by small cohorts, heterogeneous formulations, and short follow-up. Head-to-head trials against oral dutasteride, standardized pharmacokinetic data on scalp versus systemic concentrations, and longer-term safety studies are lacking. Because much prescribing relies on compounding formulations rather than standardized commercial products, variability in potency and vehicle can affect outcomes and interpretability.
Application, Dosage, And Treatment Protocols
Typical Formulations And Concentrations Used In Studies
Topical dutasteride has been studied in alcohol-based solutions, hydroalcoholic gels, and oil-in-water emulsions. Concentrations reported in the literature range from 0.01% to 0.5% (w/v). A commonly used concentration in compounding practice is 0.1%–0.5% applied once daily, though clinicians may individualize dose and frequency based on response and tolerability.
Application Methods: Topical Solution, Gel, And Microneedling Use
Solutions and gels are applied to the affected scalp once or twice daily, usually after washing and drying. Some practitioners pair topical dutasteride with microneedling to enhance drug delivery. The microchannels created by the needle array can increase local penetration and stimulate wound-healing pathways that promote growth. Microneedling is typically performed in-clinic or at home at prescribed intervals (e.g., every 2–4 weeks) and is used as an adjunct rather than a replacement for daily topical therapy.
Frequency, Duration, And Measuring Progress
Most protocols begin with daily application and reassessment at 3 and 6 months. Patients are advised to photograph the scalp with standardized lighting and angle every 3 months and, if available, use hair counts or phototrichograms for objective tracking. If improvement plateaus after 6–12 months, clinicians may consider combination therapies or escalating to oral therapy.
Safety, Side Effects, And Systemic Absorption
Local Side Effects: Scalp Irritation And Contact Reactions
Topical formulations can cause local adverse events: transient erythema, itching, dryness, and contact dermatitis, especially with alcohol-based vehicles. Switching to a buffered gel or using a lower-frequency application often mitigates irritation. Patch testing can help identify allergic responses in individuals with sensitivity.
Systemic Absorption Risks And Hormonal Effects
Although topical delivery aims to limit systemic exposure, measurable systemic absorption has been observed in some pharmacokinetic studies. Serum DHT suppression with topical dutasteride is generally lower than oral dosing, but not universally negligible; absorption depends on concentration, vehicle, application area, and skin integrity. Because dutasteride is a potent anti-androgen, even small systemic exposures raise theoretical risks of sexual dysfunction or mood changes. Reported systemic side effects in topical studies are fewer and milder than with oral therapy, but underpowered safety data mean residual risk cannot be dismissed.
Contraindications, Pregnancy Risk, And Special Populations
Dutasteride is teratogenic with respect to male fetal development: topical use is contraindicated in pregnant women, and those who may become pregnant, and women of childbearing potential should avoid handling the product. Children should not use dutasteride. Caution is advised in patients with liver disease due to hepatic metabolism.
Drug Interactions And Monitoring Recommendations
Dutasteride is metabolized by hepatic enzymes; therefore, potential interactions exist with strong CYP3A4 modulators, although clinically significant interactions from topical use are unlikely. Clinicians often monitor symptoms, consider baseline and periodic sexual health screening, and reserve serum hormone testing for cases with concerning signs of systemic exposure.
Combining Topical Dutasteride With Other Hair Treatments
Synergy With Minoxidil, Ketoconazole, And Low-Level Laser Therapy
Combining topical dutasteride with topical minoxidil addresses two complementary mechanisms: anti-androgenic blockade of follicular miniaturization and minoxidil-driven vasodilation and anagen stimulation. Ketoconazole shampoos (1%–2%) may reduce local inflammation and have modest anti-androgenic effects that support combination therapy. Low-level laser therapy (LLLT) offers noninvasive follicle stimulation and can be layered into a multimodal regimen for incremental benefit.
Procedural Adjuncts: Microneedling, PRP, And Clinic-Based Treatments
Microneedling can enhance topical absorption and stimulate growth factors. Platelet-rich plasma (PRP) and clinic-based procedures such as fractional lasers or hair transplantation remain options for patients needing procedural escalation. These procedures are frequently combined with topical regimens pre- and post-treatment to optimize graft survival and stimulate native follicles.
When To Escalate To Oral Therapy Or Specialist Referral
If topical therapy fails to produce meaningful improvement after 6–12 months, or if systemic signs suggest inadequate local control, escalation to oral dutasteride or finasteride may be considered. Referral to a board-certified dermatologist or hair specialist is warranted for diagnostic uncertainty, atypical hair loss patterns, or complex medical contraindications.
Access, Legal, And Practical Considerations
Prescription, Compounding, And Telehealth Options
Topical dutasteride is not universally available as a licensed commercial product in many markets: it’s commonly provided as a compounded prescription made by specialty pharmacies. Many clinicians now offer telehealth consultations to evaluate suitability and provide prescriptions for compounding. Because compounding practices vary, working with reputable pharmacies and clinicians is important.
Cost, Insurance, And Quality Control Of Compounded Products
Compounded topical dutasteride costs vary widely and are typically not covered by insurance. Quality control is a genuine concern: potency, sterility, and uniformity may differ between compounding pharmacies. Patients and clinicians should seek pharmacies that adhere to USP compounding standards and can provide certificates of analysis when available.
How To Discuss Topical Dutasteride With A Provider: Questions To Ask
When discussing topical dutasteride, patients should ask about the strength and vehicle proposed, expected timeline, and measurable goals, plans for monitoring side effects, alternative options (including oral therapy), and the compounding pharmacy’s quality assurances. Clear documentation of informed consent that includes pregnancy risks and potential systemic effects is prudent.
Conclusion
Topical dutasteride represents a logical, evidence-growing strategy for treating androgenetic hair loss, especially for those seeking a targeted anti-androgenic approach with potentially reduced systemic exposure. While small trials and clinical experience report encouraging gains in hair count and thickness, gaps in standardized formulations and long-term safety data remain. For practitioners and informed patients, topical dutasteride is a valuable tool within a multimodal treatment plan that might include minoxidil, procedural adjuncts, and lifestyle measures. As with any hormonally active therapy, careful patient selection, monitoring, and collaboration with reputable compounding pharmacies and clinicians are essential.